

Document Type : Original Article
Authors
1 Department of Food Science and Technology, Federal University of Technology, P.M.B.704, Akure, Nigeria
2 Department of Food Science and Technology, Joseph Ayo Babalola University Ikeji-Arakeji, Nigeria
3 Department of Food Science and Technology, Federal Polytechnic, Offa, Nigeria
Abstract
Keywords
Spontaneous “sour” dough fermentation is one of the earliest cereal fermentation methods known to man. In this technique, the dough is leavened to generate a more gaseous dough and more aerated bread. Due to its distinctively high-quality properties, the use of this dough is essential for a wide range of traditional sourdough goods, particularly for baking [1]. Lactic acid bacteria (LAB) species are often dominant in sourdough fermentation, which is a complex biological process [2].
Nowadays, consumers are highly concerned about the health aspects of the food they eat. Therefore, functional food items, such as sourdough-derived foods, are becoming increasingly popular due to their health benefits. Successful food products, on the other hand, must meet customers’ expectations for safety and optimal sensory characteristics [3]. Thus, Sourdough bread production technology is rapidly developing in recent years to obtain enhanced flavor, texture, and shelf life.
Cereals are a significant source of protein, dietary fiber (DF), vitamins, and bioactive substances. The outer layers of the grains contain the highest concentrations of the main health-protective substances, including oligosaccharides, phytochemicals, and DF [4]. According to several studies, the Western diet contains less than the daily recommended intake (25 to 30 g) of DF, and this has led to an excessive number of diseases, ranging from dental caries to obesity [5]. Therefore, wheat, barley, and oat brans have been incorporated in doughs to increase DF intake [6] and for the added nutritional value [7-9].
Fonio (Finger millet) is produced in Asia, Africa, and India and it is also known as fundi, findi, hungry rice, and Asian millet [10]. This cereal crop is resistant to viruses and pests with similar nutritional makeup compared to other cereal crops. Fonio is prepared and eaten in different indigenous forms such as tuo (tuwo i.e. in form of boiled rice with stew), djouka, couscous, gwete, acha-jollof, and kunuacha. Although it is considered native to Western Africa, Fonio attracted worldwide interest during the European Acha Project, which was run by the French Agricultural Research Center for International Development [11]. Since then, studies have demonstrated that the germination and fermentation of millets significantly lower the levels of anti-nutrients (such as phytate and tannin), promote efficient protein and starch hydrolysis, and increase mineral bioavailability in food products [12]. Furthermore, studies have reported that finger millets contain 5-8% protein, 1-2% ether extractives, 65-75% carbohydrates, 15-20% dietary fiber, 2.5-3.5% minerals, and are rich in polyphenols and calcium with high methionine content [13]. Therefore, fonio food products can assist in the management of diabetes, cardiovascular, celiac, cancer, aging, microbial and inflammatory diseases [1][11].
Lactic acid bacteria (LAB) have been used in food production for more than 6000 years (since, at least, 4000 BC), especially in dairy products [14]. These microorganisms dominated fermented plant products such as cereals, grains, and millet [15][16]. The lactobacilli are gastrointestinal tract (GIT) microorganisms necessary for maintaining and possessing probiotic activities in human digestion [17]. LAB-fermented foods are easily digestible and broken down in addition to their high antimicrobial substances content which assists in the prevention and inhibition of pathogenic bacterial growth [15]. Additionally, LAB presence in food is reported to reduce serum glucose, increase antioxidant activity, and alleviate cardiovascular and diabetes disorders [16][18].
Taking into consideration the previously mentioned points, choosing the right LAB species might be an influential factor in sourdough production. However, there is no available information regarding the influence of bacterial species type (Lactobacillus fermentum and L. delbrueckii) in the fermentation of fonio. Therefore, the goal of the current study is to determine how different starter bacteria can influence the chemical and pasting characteristics of fonio.
White fonio (Acha) (Digitaria exili) and black fonio (Iburu) (Digitaria iburua) harvested within 6–8 weeks were obtained from a farm in Kaduna State, Nigeria. The fermenting microorganisms were obtained from Bato Laboratory in Lagos State, Nigeria. The fonio seeds were verified by the Crop, Soil, and Pest Management Department of the Federal University of Technology, Akure, Nigeria.
Chemicals of analytical grade were purchased from Sigma-Aldrich (Germany and USA). All the solutions were prepared with Deionized water. All the microbiological media such as Potato Dextrose Agar (PDA), deMan, Rogosa, Sharpe Agar (MRS), and Nutrient agar were obtained from Biotech (USA).
The lactic acid bacteria were isolated using standard microbiological techniques. The isolates’ identities were verified using Bergery’s manual of identification [19] by examining the morphological and cultural characteristics of the colony formation. The identified Lactobacillus plantarum and Lactobacillus fermentum were isolated from deMan Rogosa Sharpe (MRS) broth and kept at 10 °C. Each Lactobacilli sp. broth culture was washed in 0.1 M potassium phosphate buffer three times before being centrifuged at 6000 rpm for 10 min using a refrigerated centrifuge (ThrmoFisher Scientific Sorvall ST16, USA). The cells were then re-suspended in another 20 mL of sterile 0.1 M potassium phosphate (pH 7.0), and standardized using a spectrophotometer (Model ST-VS-721, Hongkong) at a wavelength of 600 nm according to [20].
Both white and black seeds were sorted and cleaned to remove extraneous particles. The seeds were washed, drained, and dried in an oven for 8 h at 50°C. Subsequently, the dried seeds were grounded into flour and sieved separately to obtain fine and smooth flouraccording to [21]. The prepared flours were kept in hermetically sealed containers for future use.
Sourdough was developed using the paste of fonio flours (white or black) as a substrate in a ratio of 1:1 (flour: water) and producedaccording to the method described by [22]. Initial Fermentation of the substrate with L. fermentum and L. delbrueckii as single and combined (1:1) starter was carried with the addition of water for 48 h at 25°C. After that, the sourdough was left to ferment for another 8 h at 25 °C before being freeze-dried and stored.
For microbial tests, diluted saline suspensions of the sourdoughs were prepared. For that purpose, 0.85 g of sodium chloride was dissolved in 100 ml of distilled water. Then, 9 ml of the solution was added into MacConkey bottles, which were autoclaved at 121°C for 15 min. The bottles were cooled after sterilization, and one gram of sourdough was added to make 10-1 dilution. 1 ml from dilution 10-1 was taken into another bottle to make 10-2 dilution and again to make 10-3 dilution. Petri dishes were then prepared by dispensing 1 ml of the diluted sourdough suspensions into a sterilized petri dish.
Nutrient agar (NA) of 2.8 g was added to 100 ml of distilled water and autoclaved at 121°C for 15 min. Already cooled (45°C) molten agar was poured on prepared Petri dishes, rocked for even distribution, allowed to solidify, inverted, and incubated at 37 °C for 24 h before the colony count was inspected using a colony counter.
Potato Dextrose Agar (PDA) (3.9 g) was added to 100 ml of distilled water and autoclaved at 121°C for 15 min. Already cooled (45°C) molten agar was poured on prepared Petri dishes, rocked for even distribution, allowed to solidify, inverted, and incubated at 25 °C for 72 to 120 h before the colony count was inspected using a colony counter.
deMan Rogosa Sharpe (MRS) (6.3 g) was added to 100 ml of distilled water and autoclaved at 121°C for 15 min. Already cooled (45°C) molten agar was poured on prepared Petri dishes, rocked for even distribution, allowed to solidify, inverted, and incubated at 37 °C for 24 to 48 h in a gas jar with a lighted candle to create anaerobic condition before the colony count was inspected using colony counter.
Proximate content (%) for crude fat, protein, fiber, moisture, and ash of the microbial fermented sourdough was determined as described in [23]. Carbohydrate (CHO) content was determined as the remaining of other proximate compositions:
Carbohydrate (%) = 100- (Fat + Protein + Fiber + Moisture)
The energy value was calculated as:
Energy value (kJ/100g) = (Crude fat × 9) + (CHO × 4) + (Crude protein × 4)
Determination of oxalate, tannin, phytate, and saponin of microbial fermented sourdough samples was done according to [12][23]
The mineral composition of microbial fermented sourdough samples was determined using Atomic Absorption Spectrophotometer (AAS Model SP9) for calcium (Ca), iron (Fe), and zinc (Zn), and using flame photometer (FF902 UK) for potassium (K) and sodium (Na), while AOAC standard method was used to quantify NaCl as described by [12].
Free radical scavenging ability
DPPH (2,2-diphenyl-1-picrylhydrazyl) method was used to estimate the free radical scavenging capacity of the produced sourdoughs [24]. First, 0.4 mM DPPH methanolic solution of 1 mL was added to 1 mL of flour extract (1:9 dough:water w/v). The assays were then incubated in the dark at room temperature for 30 min. The absorbance of the left (unscavenged) DPPH was determined at 516 nm. DPPH scavenging ability of flour samples was then estimated compared to prepared standards, while ethylene diamine tetraacetic acid (EDTA) serves as a control standard.
Total phenolic content determination
The total phenolic content of each microbial fermented sourdough was determined according to the method of [25]. Each flour extract (1:9 dough: water w/v) was diluted with 2.5 mL of 10% Folin–Ciocalteau reagent (vol/vol) before the addition of 2.0 mL 7.5% sodium carbonate and incubation at 45 °C for 40 min. The absorbance of each sample was measured with a spectrophotometer at 765 nm compared to gallic acid standards. Then, the total phenolic content was estimated and stated in gallic acid equivalent (GAE).
Determination of total flavonoid content
The method described by [26] was followed to determine the total flavonoid content in the prepared sourdoughs. For that purpose, 0.5 g of each of the microbial fermented sourdough was diluted with a mixture of methanol (0.5 mL), 10% AlCL3 (50 µL), 1 M potassium acetate (50 µL), and distilled water (1.4 mL). These mixtures were incubated for 30 min at 25°C and the absorbance of each sample was measured at 415 nm and compared with quercetin standards. The total flavonoid content was then estimated and stated in quercetin equivalent (QE).
Microbial fermented sourdoughs were assessed using Rapid Visco Analyzer (RVA) according to [27]. Five grams of each sourdough was added to distilled water (25 mL) in a canister. The substrates were then carried into the canister’s water surface after inserting the canister analyzer piston. The piston surface was forcefully moved shakily in undulation movement severely to achieve a smooth flour sample. The canister and its paddle were rigidly thrust to the paddle coupling for proper fitting. The sourdough samples became sticky as a result of the high thick-flowing consistency and were subjected to controlled shear and high temperature to be stabilized and cooled to show any setback throughout gelatinization. The watery mixture was reheated to 95 °C from 50 °C before cooling back to 50 °C for 3 min. The pasting measurements were determined using Thermocline software (Warriewood, Australia).
The experiments were carried out in triplicates using a completely randomized design and the data obtained were subjected to analysis of variance (ANOVA) and means were separated using New Duncan’s Multiple Range Tests (NDMRT) using Statistical Package for Social Science (SPSS) version 21.0 (IBM Inc., USA) with data presented as Mean ± Standard deviations.
The total bacterial count ranged from 1.73×105 to 8.86×105 cfu/g, with the black fonio sample fermented with Lactobacillus fermentum (BFF) having the highest (8.86×105cfu/g) and white fonio fermented with Lactobacillus delbrueckii (WFD) having the least counts (1.73×105 cfu/g) (Table 1). It was also observed that sourdough fermented with L. fermentum had higher bacterial count compared to those fermented with L. delbrueckii. The total yeast count obtained ranged from 1.74×105 to 7.10×105 cfu/g. Black fonio sample fermented with L. delbrueckii (BFD) had the highest yeast count (7.10×105 cfu/g), while BFF had the least (1.74×105 cfu/g). LAB count ranged from 3.36×105 in BFF to 5.53×105 cfu/g in WFC. The difference in LAB could be attributed to the different Lactobacillus strains used for fermentation, as it appears that L. delbrueckii and control fermentation (no starter) made the medium suitable for LAB growth. On the other hand, L. fermentum starter resulted in a higher yeast count compared to L. delbrueckii. In general, the microbial loads of the samples were high, similar to other reports [27]. This could be a result of inherent microorganisms present in the fonio seeds as well as the environmental conditions during and after fermentation.
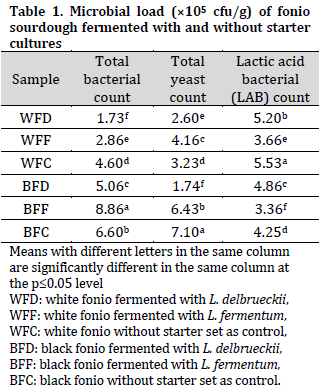
There was a significant difference in the moisture content of the samples which ranged from 12.78% in BFC to 15.62% in WFF. The differences in moisture content of the sourdough samples could be due to differences between the two fonio seed types as well as the difference in the fermentation starter specie, as L. fermentum samples scored higher moisture content followed by L. delbrueckii and control samples (Table 2). In fact, prolonged fermentation time might increase moisture content in LAB-fermented samples compared with the naturally fermented samples (control), which was previously observed in LAB-fermented maize flour [29]. Since a lower moisture content is preferable when aiming to prolong the storability of any food product, the currently observed moisture contents were within the acceptable levels [10] with no adverse effect on the quality attribute of the product.
The protein content of the samples varied from 12.07% to 18.31%. It was observed that the samples fermented with L. fermentum had higher protein contents compared to those fermented with L. delbrueckii regardless of the fonio variety (Table 2). This could be a result of the metabolic activities and high microbial density of L. fermentum,which increased protein synthesis during fermentation [30][31]. This finding is in line with the findings of [32] who reported that lyophilized cultures of microorganisms added to cereal grains increase their basic nutritive protein quality.
The fiber content of the samples ranged from 0.14% in WFC sample to 1.65% in WFD sample, which highlights the importance of directed fermentation in enhancing fiber content [33]. The fat content of the samples ranged from 11.11% in BFD to 29.22% in WFD. Overall, white fonio had a higher fat content when compared to black fonio, which is in agreement with previous reports [34]. The highest carbohydrate content was observed in BFD sample (60.18%) which might be attributed to the low microbial metabolic activity of L. delbrueckii in utilizing the carbohydrate in black fonio [35]. On the other hand, the use of L. fermentum and L. delbrueckii decreased the carbohydrate content of white fonio, which might refer to white fonio carbohydrates being more available for fermentation.
The ash content of the samples ranged from 0.1% to 0.98%. However, a previous report showed similar ash levels in white and black fonio [34]. Therefore, the observed differences in the ash content could be merely attributed to the differences in other compositions between the studied doughs. As for energy value, there was no significant difference in samples BFC and BFD, while WFD scored the highest (484.78 kJ/100g) among tested doughs.
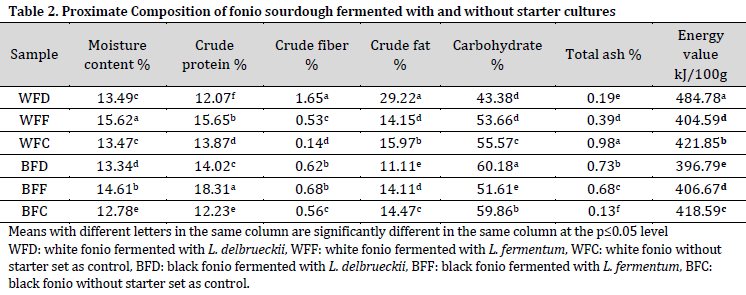
It was observed that the phytate content of sample BFF (2.74 mg/g) was significantly lower than that of the control (BFC) which scored 5.76 mg/g. This observation was similar to the findings reported by[35]that the phytic acid of fermented pearl millet was reduced from 858.4 mg/ 100 g in the raw pearl millet to 380.3 mg/100 g during the development of bread. In fact, fermentation and heat treatment are known to reduce phytate content [36]. However, this effect was not observed in WFD, WFF, or WFC, when compared to the control. Therefore, flour type and fermentation starter can both attribute to the reduction of phytate during fermentation.
Overall, oxalate, tannin, and saponin contents were lower in black fonio doughs when compared to white fonio doughs. In fact, saponin content in white fonio doughs was 5-10 times higher than their black fonio counterparts (Table 3). This means that fonio type was the determinant factor in the anti-nutritional substances content of the tested doughs.
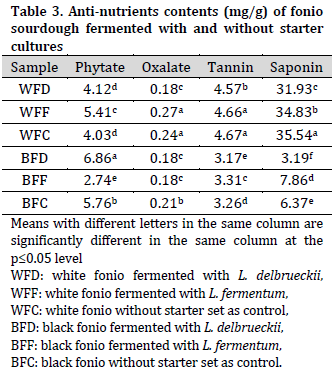
Overall, white fonio samples had higher iron (Fe), sodium (Na), and calcium (Ca) contents, while black fonio samples had higher potassium (K) content. Therefore, Na/K ratio was higher in white fonio samples compared to black fonio. On the other hand, similar zinc (Zn) levels were observed across all the samples (Table 4).
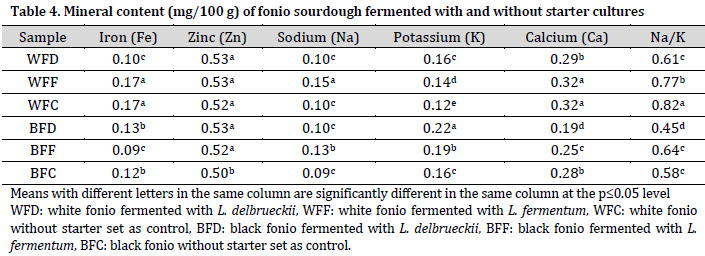
The recommended daily allowance (RDA) for Fe is 10 mg [37]. This implies that fonio flour is a poor source of iron compared to other flours, such as sorghum and soy [38]. On the other hand, the currently observed sodium levels are higher than those reported by [39].
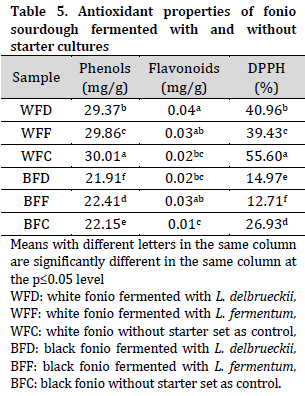
Overall, higher phenol content and DPPH levels (lower free radical scavenging ability) were observed in white fonio when compared to their black fonio counterparts. While no major differences were observed in flavonoid contents between the two types (Table 5). It was noted that fonio doughs fermented with starter LAB (except for BFF) had lower phenolic content than that of control samples. This observation contradicts previous reports that phenols concentration increases as a result of LAB-mediated fermentation of cereals and pseudo-cereal flours [41-43]. However, LAB-fermented samples had a higher free radical scavenging ability (lower unscavenged DPPH%) compared to control doughs. It is also noted that L. fermentum doughs had a higher free radical scavenging ability compared to L. delbrueckii doughs. The higher free radical scavenging ability of LAB-fermented doughs highlights the antioxidant capabilities and health benefits of directed fermentation in sourdough.
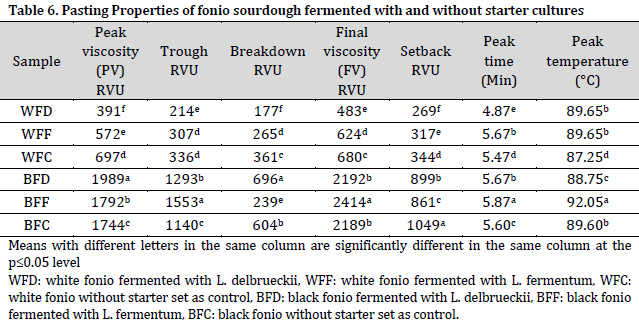
The peak viscosity (PV), trough, final viscosity (FV), and setback values were higher in black fonio sourdoughs compared to those of white fonio. PV is an index for the ability of starch-based flours to swell freely before their physical breakdown [9][45]. The currently obtained PV values are higher than those reported by [44] on chinchin production from millet and wheat. Trough viscosity is the minimum viscosity value in the constant temperature phase of the RVA pasting profile which measures the ability of the paste to withstand breakdown during cooling [46]. Therefore, white fonio samples, especially WFD, would not be able to withstand breakdown during cooling due to their low low trough viscosity values, which may lead to poor product characteristics when being baked. Additionally, the low final viscosity (FV) of sample WFD could be attributed to the lower degree of re-association between the starch molecules on subsequent cooling and cooking to form a viscous paste, since FV usually measures the stability of the cooked paste [46]. On the other hand, BFF had the highest FV value which is similar to a more stable wheat and millet composite flour in a previous report [44]. Additionally, the obtained pasting temperatures are comparable to those reported by [46]. The pasting temperature is an indicator of the minimum cooking temperature of the sample. Therefore, high pasting temperature brings about high-water binding capacity and high gelatinization tendency, which normally results in low swelling properties of any starch-based flour due to the high association between starch granules [46]. This may influence the energy cost and storability of other components in the formulation, as more energy will be required to initiate starch gelatinization in flour viscosity during heating.
The sourdoughs of white and black fonio seeds fermented with L. delbrueckii and L. fermentum as starter cultures obtained various nutritional qualities. Therefore, LAB fermented fonio sourdoughs are of high health and nutritional importance. Further research is needed to investigate the processing of these flours in baking and to provide in-depth information regarding the acceptability and rheological behavior of these doughs.
The authors wish to acknowledge the Departments of Food Science and TechnologyLaboratories of the Federal University of Technology, Akure for the assistance offered in the processing of raw materials and running the analyses.
Conflict of interest statement
The authors declared no conflict of interest.
Funding statement
The authors declared that no funding was received in relation to this manuscript.
Data availability statement
The authors declared that all related data are included in the article.